|
Although auditory cortex is not required for conventional PPI, it is essential for the detection of brief gaps (≤32 ms in duration) ( Ison et al., 1991 Kelly et al., 1996 Bowen et al., 2003 Threlkeld et al., 2008 Masini et al., 2012 Weible et al., 2014b, 2020b). Both ascending and descending auditory pathways in the brainstem, midbrain, and cortex contribute to PPI (for review, see Li et al., 2009 Lauer et al., 2017). PPI is a fundamental form of sensorimotor gating in the brain, which is impaired in a number of neuropsychiatric disorders ( Koch, 1999). Here we measured gap detection using a common variant of pre-pulse inhibition (PPI) of the acoustic startle response, in which silent gaps inserted into continuous background noise act as cues to attenuate the startle reflex. Gap detection serves as a model for temporal processing challenges faced by the auditory system, such as the detection and identification of speech sounds, species-specific vocalizations, or other events signaled by acoustic fluctuations in the environment ( Plomp, 1964 Green, 1985). Here we focus on gap detection, a behavioral temporal processing paradigm that involves primary auditory cortex, and that provides a robust and quantitative behavioral output. Pursuit of this goal has benefited tremendously from advances in the ability to manipulate and record from identified cell types in behaving animals (for review, see Adesnik and Naka, 2018). Understanding how cortical circuitry contributes to sensory perception and behavioral output is one of the central goals of systems neuroscience. These data demonstrate that cortical Gpr26 neurons are both necessary and sufficient for top–down modulation of the acoustic startle reflex, and are thus likely to be involved in sound detection. 
Photosuppression of these cells reduced neural responses to gaps throughout cortex, and impaired behavioral gap detection. Photostimulation of these cells, which were responsive to gaps in noise, was sufficient to attenuate the startle reflex. We used a Gpr26-Cre driver line, which we found expressed predominantly in layer 4 of auditory cortex. 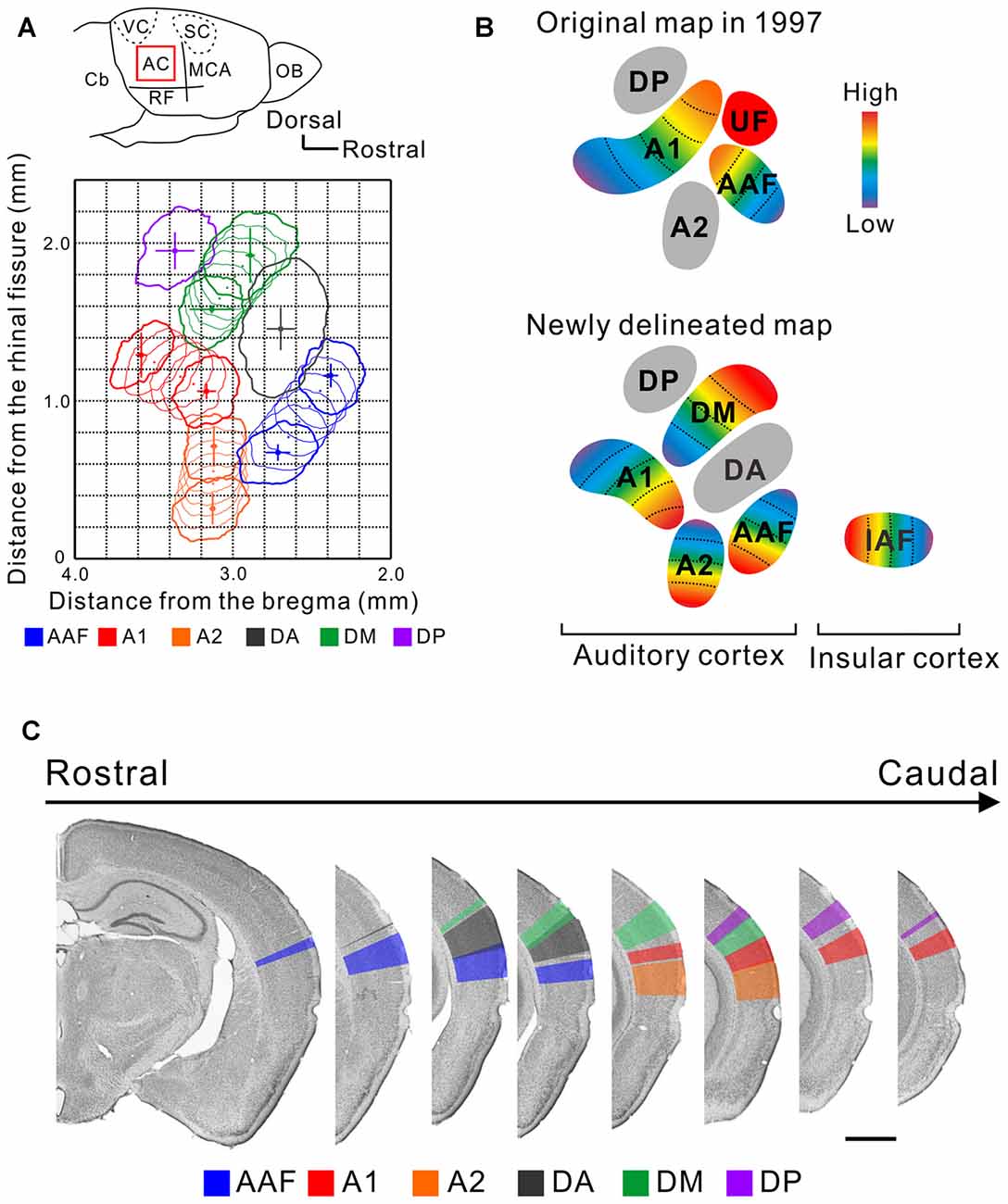
We measured sound detection using a common variant of pre-pulse inhibition of the acoustic startle response, in which a silent gap in background noise acts as a cue that attenuates startle. Here we tested the role of a genetically identified population of layer 4 auditory cortical neurons in sound detection. Spiking in auditory cortical neurons is critical for sound detection, but the causal roles of specific cell types and circuits are still mostly unknown. This data will allow us to classify and map the locations of neurons in the auditory cortex that are responsible for processing changes in sound.A fundamental task faced by the auditory system is the detection of events that are signaled by fluctuations in sound. With optical access to the auditory cortex, we are able to use in-vivo two-photon calcium imaging to evaluate sound-evoked responses of hundreds of auditory cortical neurons. Here, we report an updated protocol for cranial window implantation over the auditory cortex for use in in-vivo two-photon calcium imaging. 
However, the auditory cortex is not easily accessible during surgery due its lateral location and large muscles and arteries around the ears. To image activity at the neuronal level, optical access is achieved via implantation of a cranial window. To better understand how the brain processes sounds, we will use this imaging technique to measure the activity of neurons in the auditory cortex when an awake mouse is presented with changes in sounds. In-vivo two-photon calcium imaging is a powerful tool that enables us to measure the activity of hundreds of individual neurons simultaneously.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
